Anyone can consider participating in a clinical trial. The type of clinical trial in which you can participate will depend on the phase of the trial and the kinds of participants needed to test a new treatment. Some trials require participants with a specific medical condition. Participation in these trials often requires referral from a doctor, while others require participants with no medical conditions. Learn more about the phases of a clinical trial, here:
Anyone can consider participating in a clinical trial. You can volunteer to participate in a clinical trial if you are healthy or if you have a medical condition.
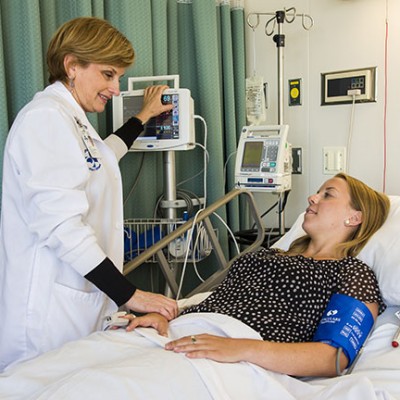
And it is the illness or medical condition studied in the clinical trial, you may be eligible to participate.
For you to make the best decision about being in a clinical trial, you need to know as much as possible. Ask your healthcare or clinical trial team any and all questions you may have.
All clinical trials follow a process and are performed according to a protocol. You will:
You will not be asked to participate in a clinical trial unless your doctor, healthcare and clinical trial team think this is a good option for you to consider. The aim of clinical trials is for the benefits to be greater than the risks. By participating in a clinical trial, you may be:
When you participate in a clinical trial you are closely watched by the clinical trial team to make sure that any side effects are carefully monitored and managed, that the treatment is given the right way, and that you do your part to follow the protocol.
There are a lot of people who are needed to run a clinical trial. They make sure everything goes according to plan and carefully monitor all participants. The people listed below will often be involved in clinical trials. A brief explanation of each person’s role is given.
Participant – A clinical trial simply isn’t possible without the generosity of the people who volunteer to participate.
Principal Investigator (PI) – The PI supervises the clinical trial across all the sites that are part of the clinical trial. This person is highly qualified (often a medical doctor or a dentist) with additional training in the area of clinical trials.
Clinical Research Coordinator – You will likely have the most contact with one or more coordinators. Coordinators ensure that participants understand how the study works and are often responsible for supporting them through a variety of activities – from completing the informed consent form to filling out trial questionnaires and studies. Coordinators may also make sure trial visits, tests and procedures are scheduled on time. They can also provide education about the clinical trial to staff and healthcare providers who may refer patients to the study.
Other Members of the Clinical Trial Team – This might include patient partners, physician assistants, physiotherapists, nurses, pharmacists, psychologists, social workers, and any other healthcare professional that might need to help care for participants throughout a clinical trial.
Sponsor – The person, company or organization that starts and manages a clinical trial, and if required, applies for permission (e.g., from Health Canada) to conduct the trial. The sponsor may or may not provide financing for the clinical trial.
Research Ethics Board (REB)– The REB is an independent group that reviews the ethics (the right and wrong) of a clinical trial and ensures that participants in the clinical trial are protected.
The treatment may not have an effect or work no better than your current treatment.
These may range from minor and unpleasant to serious and even life threatening.
You may or may not be able to continue your current treatment during the clinical trial.
You may need to go to several appointments, have more tests, or keep a daily clinical trial diary.
Participating in a clinical trial is voluntary and completely up to you. When you are invited to participate (and even after you have started the clinical trial) you have the right to:
Here are some trustworthy resources to help you find a clinical trial.
Canadian Resources:
International Resources:
You may also find out more about how to be in a clinical trial by asking your doctor or another member of your healthcare team.